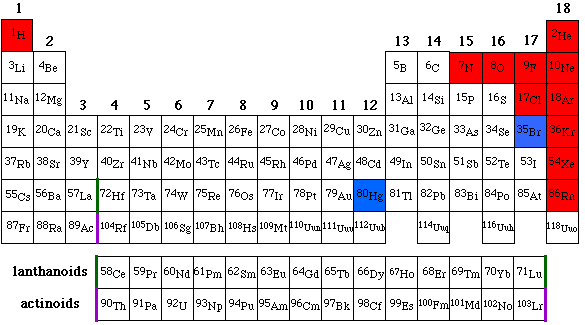
 |
||||||
|
||||||
| Hydrogen and the alkali metals |
| elements with a s1 configuration outside a non-oxidizable core |
| Hydrogen |
Atomic configuration: 1s1 |
Atomic mass: 1.0079 |
Pauling electronegativity: 2.1 |
Common oxidation states: ±1
|
Ionization energy: 1312 kJ mol-1 |
Electron affinity: 72 kJ mol-1 |
Melting point: 14 K |
Boiling point: 20 K |
| identified in 1766 by H. Cavendish from action of acid on metal |
| Lithium |
Atomic configuration: [He] 2s1 |
Atomic mass: 6.941 |
Pauling electronegativity: 0.98 |
Common oxidation states: +1 |
Ionization energy: 520 kJ mol-1 |
Electron affinity: 60 kJ mol-1 |
Melting point: 454 K |
Boiling point: 1615 K |
| identified in 1817 by Arfvedson and isolated from mineral petalite |
| Sodium |
Atomic configuration: [Ne] 3s1 |
Atomic mass: 22.9898 |
Pauling electronegativity: 0.93 |
Common oxidation states: +1 |
Ionization energy: 496 kJ mol-1 |
Electron affinity: 53 kJ mol-1 |
Melting point: 371 K |
Boiling point: 1156 K |
| isolated by Davy in 1807 by electrolysis of NaOH days after discovery of K |
| Potassium |
Atomic configuration: [Ar] 4s1 |
Atomic mass: 39.0983 |
Pauling electronegativity: 0.82 |
Common oxidation states: +1 |
Ionization energy: 419 kJ mol-1 |
Electron affinity: 49 kJ mol-1 |
Melting point: 337 K |
Boiling point: 1033 K |
| isolated by Davy in 1807 by electrolysis of molten KOH when 29 years old |
| Rubidium |
Atomic configuration: [Kr] 5s1 |
Atomic mass: 85.4678 |
Pauling electronegativity: 0.82 |
Common oxidation states: +1 |
Ionization energy: 403 kJ mol-1 |
Electron affinity: 47 kJ mol-1 |
Melting point: 313 K |
Boiling point: 961 K |
| identified in 1861 by Bunsen and Kirchoff in mineral lepidolite |
| Caesium |
Atomic configuration: [Xe] 6s1 |
Atomic mass: 132.9054 |
Pauling electronegativity: 0.79 |
Common oxidation states: +1 |
Ionization energy: 376 kJ mol-1 |
Electron affinity: 46 kJ mol-1 |
Melting point: 302 K |
Boiling point: 944 K |
| identified in 1860 by Bunsen and Kirchoff in mineral spa waters |
| Francium |
Atomic configuration: [Rn] 7s1 |
Atomic mass: (223) |
Pauling electronegativity: 0.71 |
Common oxidation states: +1 |
Ionization energy: ~375 kJ mol-1 |
Electron affinity: ~44 kJ mol-1 |
Melting point: 300 K |
Boiling point: 950 K |
| identified in 1939 by Percy from radioactive decay of Ac |
| The alkaline earths |
| elements with a s2 configuration outside a noble gas core |
| Beryllium |
Atomic configuration: [He] 2s2 |
Atomic mass: 9.0122 |
Pauling electronegativity: 1.57 |
Common oxidation states: +2 |
Ionization energy: 899 kJ mol-1 |
Electron affinity: 0 kJ mol-1 |
Melting point: 1289 K |
Boiling point: 2472 K |
| identified in 1798 by Vauquelin in beryllia from beryl and emerald |
| Magnesium |
Atomic configuration: [Ne] 3s2 |
Atomic mass: 24.305 |
Pauling electronegativity: 1.31 |
Common oxidation states: +2 |
Ionization energy: 738 kJ mol-1 |
Electron affinity: 0 kJ mol-1 |
Melting point: 650 K |
Boiling point: 1090 K |
| known since antiquity: isolated by Davy (1808) by electrolysis |
| Calcium |
Atomic configuration: [Ar] 4s2 |
Atomic mass: 40.08 |
Pauling electronegativity: 1.00 |
Common oxidation states: +2 |
Ionization energy: 590 kJ mol-1 |
Electron affinity: 0 kJ mol-1 |
Melting point: 842 K |
Boiling point: 1494 K |
| known since antiquity: isolated by Davy (1808) by electrolysis |
| Strontium |
Atomic configuration: [Kr] 5s2 |
Atomic mass: 87.62 |
Pauling electronegativity: 0.95 |
Common oxidation states: +2 |
Ionization energy: 550 kJ mol-1 |
Electron affinity: 0 kJ mol-1 |
Melting point: 769 K |
Boiling point: 1382 K |
| identified by Crawford in 1790 and confirmed by Hope in 1791 |
| Barium |
Atomic configuration: [Xe] 6s2 |
Atomic mass: 137.33 |
Pauling electronegativity: 0.89 |
Common oxidation states: +2 |
Ionization energy: 503 kJ mol-1 |
Electron affinity: 0 kJ mol-1 |
Melting point: 729 K |
Boiling point: 1805 K |
| known since the 17th Century and isolated by Davy in 1808 |
| Radium |
Atomic configuration: [Rn] 7s2 |
Atomic mass: 226.03 |
Pauling electronegativity: 0.90 |
Common oxidation states: +2 |
Ionization energy: 509 kJ mol-1 |
Electron affinity: -
|
Melting point: 700 K
|
Boiling point: (1700 K) |
| isolated by the Curies in 1898 by processing tonnes of pitchblende |
| Group 3 |
| elements with a s2d1 configuration outside a noble gas core |
| Scandium |
Atomic configuration: [Ar] 4s23d1 |
Atomic mass: 44.956 |
Pauling electronegativity: 1.3 |
Common oxidation states: +3 |
Ionization energy: 633 kJ mol-1 |
Electron affinity: 18 kJ mol-1 |
Melting point: 1539 K |
Boiling point: 2748 K |
| identified in 1879 by Nilsen and isolated in 1937 by electrolysis |
| Yttrium |
Atomic configuration: [Kr] 5s24d1 |
Atomic mass: 88.906 |
Pauling electronegativity: 1.2 |
Common oxidation states: +3 |
Ionization energy: 600 kJ mol-1 |
Electron affinity: 30 kJ mol-1 |
Melting point: 1530 K |
Boiling point: 3264 K |
| oxide separated in 1843 by Mosander from mineral yttria |
| Lanthanum |
Atomic configuration: [Xe] 6s25d1 |
Atomic mass: 138.906 |
Pauling electronegativity: 1.1 |
Common oxidation states: +3 |
Ionization energy: 538 kJ mol-1 |
Electron affinity: 48 kJ mol-1 |
Melting point: 920 K |
Boiling point: 3420 K |
| oxide extracted in 1839 by Mosander from cerium nitrate |
| Actinium |
Atomic configuration: [Rn] 7s26d2 |
Atomic mass: 227.028 |
Pauling electronegativity: 1.1 |
Common oxidation states: +3 |
Ionization energy: 499 kJ mol-1 |
Electron affinity: -
|
Melting point: 817 K
|
Boiling point: 2470 K |
| identified in 1899 by Debierne in urannium minerals |
| Group 4 |
| elements with a s2d2 configuration outside a noble gas core |
| Titanium |
Atomic configuration: [Ar] 4s23d2 |
Atomic mass: 47.88 |
Pauling electronegativity: 1.5 |
Common oxidation states: +2, +3, +4 |
Ionization energy: 659 kJ mol-1 |
Electron affinity: 8 kJ mol-1 |
Melting point: 1667 K |
Boiling point: 3285 K |
| identified in 1791 by Cornish vicar Gregor in sand from the river Helford |
| Zirconium |
Atomic configuration: [Kr] 5s24d2 |
Atomic mass: 91.22 |
Pauling electronegativity: 1.4 |
Common oxidation states: +3, +4 |
Ionization energy: 640 kJ mol-1 |
Electron affinity: 41 kJ mol-1 |
Melting point: 1857 K |
Boiling point: 4200 K |
| discovered in 1789 by German chemist Klaproth in rutile ore |
| Hafnium |
Atomic configuration: [Xe] 4f146s25d2 |
Atomic mass: 178.49 |
Pauling electronegativity: 1.3 |
Common oxidation states: +3, +4 |
Ionization energy: 659 kJ mol-1 |
Electron affinity: 0 kJ mol-1 |
Melting point: 2222 K |
Boiling point: 4450 K |
| identified in 1922 by Coster and von Gersey using X-ray spectroscopy on zircon |
| Rutherfordium |
Atomic configuration: [Rn] 5f147s26d1 |
Atomic mass: (263) |
Pauling electronegativity: - |
Common oxidation states: - |
Ionization energy: -
|
Electron affinity: -
|
Melting point: -
|
Boiling point: - |
| synthesized in 1964 in Dubna and in 1969 in Berkeley by high energy collisions |
| Group 5 |
| elements with a s2d3 configuration outside a noble gas core |
| Vanadium |
Atomic configuration: [Ar] 4s23d3 |
Atomic mass: 50.94' |
Pauling electronegativity: 1.6 |
Common oxidation states: +2, +3, +4, +5 |
Ionization energy: 651 kJ mol-1 |
Electron affinity: 51 kJ mol-1 |
Melting point: 1915 K |
Boiling point: 3350 K |
| discovered twice in 1801 by del Rio and in 1830 by Sefstr�m |
| Niobium |
Atomic configuration: [Kr] 5s24d3 |
Atomic mass: 92.91 |
Pauling electronegativity: 1.6 |
Common oxidation states: +2, +3, +4, +5 |
Ionization energy: 652 kJ mol-1 |
Electron affinity: 86 kJ mol-1 |
Melting point: 2468 K |
Boiling point: 4758 K |
| discovered by Hatchett in 1801 and originally called columbian |
| Tantalum |
Atomic configuration: [Xe] 4f146s25d3 |
Atomic mass: 180.95 |
Pauling electronegativity: 1.5 |
Common oxidation states: +2, +3, +4, +5 |
Ionization energy: 761 kJ mol-1 |
Electron affinity: 31 kJ mol-1 |
Melting point: 2980 K |
Boiling point: 534 K |
| identified in Sweden by Ekeberg in 1802 in Finnish minerals |
| Dubnium |
Atomic configuration: [Rn] 5f147s26d3 |
Atomic mass: (262) |
Pauling electronegativity: - |
Common oxidation states: - |
Ionization energy: -
|
Electron affinity: -
|
Melting point: -
|
Boiling point: - |
| made in Dubna in 1967 and in Berkeley in 1970 by atom bombardment |
| Group 6 |
| elements with 6 valence electrons and sndm configurations |
| Chromium |
Atomic configuration: [Ar] 4s13d5 |
Atomic mass: 52.00' |
Pauling electronegativity: 1.8 |
Common oxidation states: +2, +3, +4, +5, +6 |
Ionization energy: 653 kJ mol-1 |
Electron affinity: 54 kJ mol-1 |
Melting point: 1244 K |
Boiling point: 2060 K |
| discovered in 1797 by Vauquelin in the mineral crocoite |
| Molybdenum |
Atomic configuration: [Kr] 5s14d5 |
Atomic mass: 95.94 |
Pauling electronegativity: 2.26 |
Common oxidation states: +2, +3, +4, +5 |
Ionization energy: 684 kJ mol-1 |
Electron affinity: 72 kJ mol-1 |
Melting point: 2200 K |
Boiling point: 4567 K |
| identified in 1778 by Scheele in the mineral now called molybdenite |
| Tungsten |
Atomic configuration: [Xe] 4f146s25d4 |
Atomic mass: 183.84 |
Pauling electronegativity: 2.2 |
Common oxidation states: +2, +3, +4, +5, +6 |
Ionization energy: 770 kJ mol-1 |
Electron affinity: 79 kJ mol-1 |
Melting point: 3695 K |
Boiling point: 5828 K |
| identified in 1781 by Scheele and Bergman in the mineral scheelite |
| Seaborgium |
Atomic configuration: [Rn] -
|
Atomic mass: (266)
|
Pauling electronegativity: - |
Common oxidation states: - |
Ionization energy: -
|
Electron affinity: -
|
Melting point: -
|
Boiling point: - |
| made in 1974 in the Soviet Joint Institute for Nuclear Research from Cr and Pb |
| Group 7 |
| elements with 7 valence electrons and sndm configurations |
| Manganese |
Atomic configuration: [Ar] 4s23d5 |
Atomic mass: 54.94' |
Pauling electronegativity: 1.5 |
Common oxidation states: +2, +3, +4, +5, +6, +7 |
Ionization energy: 717 kJ mol-1 |
Electron affinity: 0 kJ mol-1 |
Melting point: 1900 K |
Boiling point: 2690 K |
| isolated in 1774 by Scheele and Gahn from the mineral pyrolusite |
| Technetium |
Atomic configuration: [Kr] 5s14d6 |
Atomic mass: (98) |
Pauling electronegativity: 1.9 |
Common oxidation states: +2, +3, +4, +5, +6, +7 |
Ionization energy: 702 kJ mol-1 |
Electron affinity: 53 kJ mol-1 |
Melting point: 1900 K |
Boiling point: 2690 K |
| first artificial element, made by Perrier and Segr� in 1937 by firing neutrons at Mo |
| Rhenium |
Atomic configuration: [Xe] 4f146s25d5 |
Atomic mass: 186.21 |
Pauling electronegativity: 1.95 |
Common oxidation states: +2, +3, +4, +5, +6, +7 |
Ionization energy: 760 kJ mol-1 |
Electron affinity: 15 kJ mol-1 |
Melting point: 3180 K |
Boiling point: 5869 K |
| discovered in 1925 by Noddack, Tacke and Berg in mineral gadolinite |
| Bohrium |
Atomic configuration: [Rn] -
|
Atomic mass: (264)
|
Pauling electronegativity: - |
Common oxidation states: - |
Ionization energy: -
|
Electron affinity: -
|
Melting point: -
|
Boiling point: - |
| made in USSR in 1974 and in Darmstadt in 1981 by atom bombardment |
| Group 8 |
| elements with 8 valence electrons and sndm configurations |
| Iron |
Atomic configuration: [Ar] 4s23d6 |
Atomic mass: 55.85' |
Pauling electronegativity: 1.8 |
Common oxidation states: +2, +3, +4, +5, +6 |
Ionization energy: 763 kJ mol-1 |
Electron affinity: 16 kJ mol-1 |
Melting point: 1495 K |
Boiling point: 3100 K |
| known since prehistory and first smelted around 3000BC in Asia Minor |
| Ruthenium |
Atomic configuration: [Kr] 5s14d7 |
Atomic mass: 101.07 |
Pauling electronegativity: 2.2 |
Common oxidation states: +2, +3, +4, +5, +6, +7, +8 |
Ionization energy: 710 kJ mol-1 |
Electron affinity: 101 kJ mol-1 |
Melting point: 1495 K |
Boiling point: 3100 K |
| discovered in 1844 by Klaus in residue after Pt is dissolved in aqua regia |
| Osmium |
Atomic configuration: [Xe] 4f146s25d6 |
Atomic mass: 190.2 |
Pauling electronegativity: 2.2 |
Common oxidation states: +2, +3, +4, +5, +6, +7, +8 |
Ionization energy: 840 kJ mol-1 |
Electron affinity: 106 kJ mol-1 |
Melting point: 1495 K |
Boiling point: 3100 K |
| discovered in 1803 by Tenant in residue after Pt is dissolved in aqua regia |
| Hassium |
Atomic configuration: [Rn] -
|
Atomic mass: (269)
|
Pauling electronegativity: - |
Common oxidation states: - |
Ionization energy: -
|
Electron affinity: -
|
Melting point: -
|
Boiling point: - |
| made in 1984 in Darmstadt, Germany, by fast atom bombardment |
| Group 9 |
| elements with a 9 valence electrons and sndm configurations |
| Cobalt |
Atomic configuration: [Ar] 4s23d7 |
Atomic mass: 58.93' |
Pauling electronegativity: 1.8 |
Common oxidation states: +2, +3, +4 |
Ionization energy: 760 kJ mol-1 |
Electron affinity: 64 kJ mol-1 |
Melting point: 1495 K |
Boiling point: 3100 K |
| ores used for colour since antiquity and isolated in 1735 by Brandt |
| Rhodium |
Atomic configuration: [Kr] 5s14d8 |
Atomic mass: 102.91 |
Pauling electronegativity: 1.8 |
Common oxidation states: +1, +2, +3, +4, +5, +6 |
Ionization energy: 720 kJ mol-1 |
Electron affinity: 110 kJ mol-1 |
Melting point: 1960 K |
Boiling point: 3760 K |
| discovered in 1803 by Wollaston in residue after Pt is dissolved in aqua regia |
| Iridium |
Atomic configuration: [Xe] 4f146s25d7 |
Atomic mass: 192.22 |
Pauling electronegativity: 1.8 |
Common oxidation states: +1, +3, +4, +5, +6 |
Ionization energy: 880 kJ mol-1 |
Electron affinity: 151 kJ mol-1 |
Melting point: 2443 K |
Boiling point: 4550 K |
| discovered in 1803 by Tenant in residue after Pt is dissolved in aqua regia |
| Meitnerium |
Atomic configuration: [Rn] -
|
Atomic mass: (268)
|
Pauling electronegativity: - |
Common oxidation states: - |
Ionization energy: -
|
Electron affinity: -
|
Melting point: -
|
Boiling point: - |
| one atom made in 1982 in Darmstadt by fusing an Fe and a Bi atom |
| Group 10 |
| elements with 10 valence electrons and sndm configurations |
| Nickel |
Atomic configuration: [Ar] 3d84s2 |
Atomic mass: 58.69 |
Pauling electronegativity: 1.8 |
Common oxidation states: +2, +3, +4 |
Ionization energy: 737 kJ mol-1 |
Electron affinity: 112 kJ mol-1 |
Melting point: 1455 K |
Boiling point: 2920 K |
| used in China over 2000 years ago and isolated in 1751 by Cronstedt |
| Palladium |
Atomic configuration: [Kr] 4d10 |
Atomic mass: 106.42 |
Pauling electronegativity: 2.2 |
Common oxidation states: +2, +3, +4 |
Ionization energy: 804 kJ mol-1 |
Electron affinity: 54 kJ mol-1 |
Melting point: 1552 K |
Boiling point: 2940 K |
| isolated in 1803 by Wollaston when studying chemistry of platinum |
| Platinum |
Atomic configuration: [Xe] 4f145d96s1 |
Atomic mass: 195.08 |
Pauling electronegativity: 2.2 |
Common oxidation states: +2, +3, +4, +5, +6 |
Ionization energy: 870 kJ mol-1 |
Electron affinity: 205 kJ mol-1 |
Melting point: 1769 K |
Boiling point: 4170 K |
| used for jewellery in ancient Egypt and by the Indians of Ecuador |
| Ununnilium |
Atomic configuration: [Rn] -
|
Atomic mass: (272)
|
Pauling electronegativity: - |
Common oxidation states: - |
Ionization energy: -
|
Electron affinity: -
|
Melting point: -
|
Boiling point: - |
| one atom made in Darmstadt in 1994 by fusing a Ni and a Pb atom together |
| The coinage metals |
| elements with a d10s1d configuration outside a noble gas core |
| Copper |
Atomic configuration: [Ar] 3d104s1 |
Atomic mass: 63.55 |
Pauling electronegativity: 1.9 |
Common oxidation states: +1, +2, +3 |
Ionization energy: 745 kJ mol-1 |
Electron affinity: 118 kJ mol-1 |
Melting point: 1083 K |
Boiling point: 2570 K |
| known since 5000BC, mixed with tin in India in ~3000BC to start Bronze Age |
| Silver |
Atomic configuration: [Kr] 4d105s1 |
Atomic mass: 107.87 |
Pauling electronegativity: 1.9 |
Common oxidation states: +1, +2, +3 |
Ionization energy: 731 kJ mol-1 |
Electron affinity: 126 kJ mol-1 |
Melting point: 961 K |
Boiling point: 2155 K |
| used as currency since 3000BC in many of the classical civilizations |
| Gold |
Atomic configuration: [Xe] 4f145d106s1 |
Atomic mass: 196.97 |
Pauling electronegativity: 2.4 |
Common oxidation states: +1, +2, +3, +4, +5 |
Ionization energy: 890 kJ mol-1 |
Electron affinity: 223 kJ mol-1 |
Melting point: 1064 K |
Boiling point: 2808 K |
| used as currency by ancient societies including Egypt around 3400BC |
| Unununium |
Atomic configuration: [Rn] 5f146d107s1 |
Atomic mass: (277) |
Pauling electronegativity: - |
Common oxidation states: - |
Ionization energy: -
|
Electron affinity: -
|
Melting point: -
|
Boiling point: - |
| made in 1994 in Darmstadt by hitting Bi atoms with high energy Ni atoms |
| Group 12 |
| elements with a d10s2d configuration outside a noble gas core |
| Zinc |
Atomic configuration: [Ar] 3d104s2 |
Atomic mass: 65.38 |
Pauling electronegativity: 1.65 |
Common oxidation states: +2 |
Ionization energy: 906 kJ mol-1 |
Electron affinity: 0 kJ mol-1 |
Melting point: 693 K |
Boiling point: 1180 K |
| separation from oxide probably first achieved in India in 13th Century |
| Cadmium |
Atomic configuration: [Kr] 4d105s2 |
Atomic mass: 112.41 |
Pauling electronegativity: 1.69 |
Common oxidation states: +2 |
Ionization energy: 868 kJ mol-1 |
Electron affinity: 0 kJ mol-1 |
Melting point: 594 K |
Boiling point: 1040 K |
| observed in 1817 by Stromeyer as yellow impurity in calamine |
| Mercury |
Atomic configuration: [Xe] 4f145d106s2 |
Atomic mass: 200.59 |
Pauling electronegativity: 2.0 |
Common oxidation states: +1, +2 |
Ionization energy: 1007 kJ mol-1 |
Electron affinity: 0 kJ mol-1 |
Melting point: 234 K |
Boiling point: 630 K |
| used to extract metals by amalgamation since before 500 BC |
| Ununbium |
Atomic configuration: [Rn] 5f146d107s2 |
Atomic mass: (277) |
Pauling electronegativity: - |
Common oxidation states: - |
Ionization energy: -
|
Electron affinity: -
|
Melting point: -
|
Boiling point: - |
| made in 1996 in Darmstadt by fusing zinc and lead atoms |
| Group 13 |
| elements with a s2p1 valence configuration |
| Boron |
Atomic configuration: [He] 2s22p1 |
Atomic mass: 10.81 |
Pauling electronegativity: 2.04 |
Common oxidation states: ±3
|
Ionization energy: 801 kJ mol-1 |
Electron affinity: 27 kJ mol-1 |
Melting point: 2092 K |
Boiling point: 4002 K |
| known since antiquity and isolated in 1808 by Davy, Gay, Lussac & Th�nard |
| Aluminium |
Atomic configuration: [Ne] 3s23p1 |
Atomic mass: 26.982 |
Pauling electronegativity: 1.61 |
Common oxidation states: +3 |
Ionization energy: 578 kJ mol-1 |
Electron affinity: 43 kJ mol-1 |
Melting point: 660 K |
Boiling point: 2520 K |
| known since antiquity and isolated in 1825 by Oersted. (Called aluminum in the US) |
| Gallium |
Atomic configuration: [Ar] 3d104s24p1 |
Atomic mass: 69.72 |
Pauling electronegativity: 1.00 |
Common oxidation states: +1, +3 |
Ionization energy: 579 kJ mol-1 |
Electron affinity: 29 kJ mol-1 |
Melting point: 303 K |
Boiling point: 2205 K |
| discoved by de Boisaudran in 1875 using spectroscope and isolated from zinc blende |
| Indium |
Atomic configuration: [Kr] 4d105s25p1 |
Atomic mass: 114.82 |
Pauling electronegativity: 1.78 |
Common oxidation states: +1, +3 |
Ionization energy: 558 kJ mol-1 |
Electron affinity: 29 kJ mol-1 |
Melting point: 157 K |
Boiling point: 2073 K |
| identified in 1863 by Reich and Richter using spectroscope |
| Thallium |
Atomic configuration: [Xe] 4f145d106s26p1 |
Atomic mass: 204.383 |
Pauling electronegativity: 1.62 |
Common oxidation states: +1, +3 |
Ionization energy: 580 kJ mol-1 |
Electron affinity: 19 kJ mol-1 |
Melting point: 304 K |
Boiling point: 1473 K |
| identified in 1861/2 independently by Crookes and Lamy |
| Group 14 |
| elements with a s2p2 valence configuration |
| Carbon |
Atomic configuration: [He] 2s22p2 |
Atomic mass: 12.011 |
Pauling electronegativity: 2.5 |
Common oxidation states: ±4
|
Ionization energy: 1086 kJ mol-1 |
Electron affinity: 154 kJ mol-1 |
Melting point: 3800 K |
Boiling point: 4300 K |
| known in prehistory and identified as an element in the 18th Century |
| Silicon |
Atomic configuration: [Ne] 3s23p2 |
Atomic mass: 26.982 |
Pauling electronegativity: 1.8 |
Common oxidation states: +4 |
Ionization energy: 786 kJ mol-1 |
Electron affinity: 134 kJ mol-1 |
Melting point: 1420 K |
Boiling point: 3280 K |
| known in prehistory and isolated in 1823 by J J Berzelius |
| Germanium |
Atomic configuration: [Ar] 3d104s24p2 |
Atomic mass: 72.59 |
Pauling electronegativity: 1.8 |
Common oxidation states: +2, +4 |
Ionization energy: 761 kJ mol-1 |
Electron affinity: 119 kJ mol-1 |
Melting point: 945 K |
Boiling point: 2850 K |
| predicted by Newlands and Mendeleev and idenified in 1886 by Winkler |
| Tin |
Atomic configuration: [Kr] 4d105s25p2 |
Atomic mass: 118.69 |
Pauling electronegativity: 1.8 |
Common oxidation states: +2, +4 |
Ionization energy: 708 kJ mol-1 |
Electron affinity: 107 kJ mol-1 |
Melting point: 232 K |
Boiling point: 2623 K |
| used since at least the bronze age and mentioned in the Old Testament |
| Lead |
Atomic configuration: [Xe] 4f145d106s26p1 |
Atomic mass: 207.2 |
Pauling electronegativity: 1.9 |
Common oxidation states: +2, +4 |
Ionization energy: 715 kJ mol-1 |
Electron affinity: 35 kJ mol-1 |
Melting point: 327 K |
Boiling point: 1751 K |
| used since at least the bronze age and mentioned in the Old Testament |
| Ununquadium |
Atomic configuration: [Rn] 5f146d107s27p1 |
Atomic mass: (289) |
Pauling electronegativity: - |
Common oxidation states: - |
Ionization energy: -
|
Electron affinity: -
|
Melting point: -
|
Boiling point: - |
| ONE atom made at the Nuclear Institute in Dubna in Russia in 1998 |
| The pnicogens |
| elements with three electrons missing from a noble gas configuration |
| Nitrogen |
Atomic configuration: [He] 2s22p3 |
Atomic mass: 14.007 |
Pauling electronegativity: 3.0 |
Common oxidation states: ±3, +5
|
Ionization energy: 1402 kJ mol-1 |
Electron affinity: 17 kJ mol-1 |
Melting point: 63 K |
Boiling point: 77 K |
| discovered in 1772 by Rutherford removing oxygen from air by combustion |
| Phosphorus |
Atomic configuration: [Ne] 3s23p3 |
Atomic mass: 30.974 |
Pauling electronegativity: 2.1 |
Common oxidation states: ±3, +5
|
Ionization energy: 1012 kJ mol-1 |
Electron affinity: 720 kJ mol-1 |
Melting point: 317 K |
Boiling point: 550 K |
| isolated in 1669 by alchemist Brandt putrifying and distilling urine |
| Arsenic |
Atomic configuration: [Ar] 3d104s24p3 |
Atomic mass: 74.922 |
Pauling electronegativity: 2.0 |
Common oxidation states: +3, +5 |
Ionization energy: 947 kJ mol-1 |
Electron affinity: 78 kJ mol-1 |
Melting point: sublimes at atmospheric pressure |
Boiling point: 887 K |
| known to physicians & poisoners since at least the 5th Century BC |
| Antimony |
Atomic configuration: [Kr] 4d105s25p3 |
Atomic mass: 121.75 |
Pauling electronegativity: 1.9 |
Common oxidation states: +3, +5 |
Ionization energy: 834 kJ mol-1 |
Electron affinity: 103 kJ mol-1 |
Melting point: 904 K |
Boiling point: 1860 K |
| known since antiquity and probably isolated in 1492 by Benedictine monk Valentine |
| Bismuth |
Atomic configuration: [Xe] 4f145d106s26p3 |
Atomic mass: 208.980 |
Pauling electronegativity: 1.9 |
Common oxidation states: +3 ,+5 |
Ionization energy: 703 kJ mol-1 |
Electron affinity: 91 kJ mol-1 |
Melting point: 544 K |
Boiling point: 1837 K |
| known to alchemists by 1480 and probably well before |
| The chalcogens |
| elements with two electrons missing from a noble gas configuration |
| Oxygen |
Atomic configuration: [He] 2s22p4 |
Atomic mass: 15.998 |
Pauling electronegativity: 3.5 |
Common oxidation states: ±2
|
Ionization energy: 1314 kJ mol-1 |
Electron affinity: 141 kJ mol-1 |
Melting point: 55 K |
Boiling point: 90 K |
| discovered in 1773-4 by Scheele and Priestly but probably identified earlier |
| Sulfur |
Atomic configuration: [Ne] 3s23p4 |
Atomic mass: 32.06 |
Pauling electronegativity: 2.5 |
Common oxidation states: ±2, +4, +6
|
Ionization energy: 999 kJ mol-1 |
Electron affinity: 200 kJ mol-1 |
Melting point: 388 K |
Boiling point: 718 K |
| known since prehistory and mentioned in the Bible, by Homer, Pliny and others |
| Selenium |
Atomic configuration: [Ar] 3d104s24p4 |
Atomic mass: 78.96 |
Pauling electronegativity: 2.4 |
Common oxidation states: ±2, +4, +6
|
Ionization energy: 941 kJ mol-1 |
Electron affinity: 195 kJ mol-1 |
Melting point: 494 K |
Boiling point: 958 K |
| discovered in 1872 by Berzelius and Gahn 35 years after discovery of Te |
| Tellurium |
Atomic configuration: [Kr] 4d105s25p4 |
Atomic mass: 127.60 |
Pauling electronegativity: 2.1 |
Common oxidation states: ±2, +4, +6
|
Ionization energy: 869 kJ mol-1 |
Electron affinity: 190 kJ mol-1 |
Melting point: 723 K |
Boiling point: 1261 K |
| isolated in 1782 by M�ller von Reichenstein from Transylvania ores |
| Pollonium |
Atomic configuration: [Xe] 4f145d106s26p4 |
Atomic mass: (209) |
Pauling electronegativity: 2.0 |
Common oxidation states: ±2, +4, +6
|
Ionization energy: 813 kJ mol-1 |
Electron affinity: 183 kJ mol-1 |
Melting point: 527 K |
Boiling point: 1253 K |
| discovered in 1898 by Marie Curie by processing uranium ore |
| Ununhexium |
Atomic configuration: [Rn] 5f146d107s27p4 |
Atomic mass: (289) |
Pauling electronegativity: - |
Common oxidation states: - |
Ionization energy: -
|
Electron affinity: -
|
Melting point: -
|
Boiling point: - |
| observed by team at Berkeley in 1999 from decay of Uuo-118 |
| The halogens |
| elements with one electron missing from a noble gas configuration |
| Fluorine |
Atomic configuration: [He] 2s22p5 |
Atomic mass: 18.998 |
Pauling electronegativity: 3.98 |
Common oxidation states: -1 |
Ionization energy: 1681 kJ mol-1 |
Electron affinity: 333 kJ mol-1 |
Melting point: 54 K |
Boiling point: 85 K |
| known since antiquity and isolated in 1886 by Moissan by electrolysis |
| Chlorine |
Atomic configuration: [Ne] 3s23p5 |
Atomic mass: 35.453 |
Pauling electronegativity: 3.16 |
Common oxidation states: ±,+3,+5,+7
|
Ionization energy: 1256 kJ mol-1 |
Electron affinity: 349 kJ mol-1 |
Melting point: 172 K |
Boiling point: 239 K |
| known since antiquity and isolated in 1774 by Scheele by oxidizing HCl |
| Bromine |
Atomic configuration: [Ar] 3d104s25p5 |
Atomic mass: 79.904 |
Pauling electronegativity: 2.96 |
Common oxidation states: ±,+3,+5,+7
|
Ionization energy: 1143 kJ mol-1 |
Electron affinity: 325 kJ mol-1 |
Melting point: 266 K |
Boiling point: 332 K |
| known since antiquity and isolated in 1826 by Balard from salt marsh water |
| Iodine |
Atomic configuration: [Kr] 4d105s25p5 |
Atomic mass: 126.905 |
Pauling electronegativity: 2.66 |
Common oxidation states: ±,+3,+5,+7
|
Ionization energy: 1009 kJ mol-1 |
Electron affinity: 295 kJ mol-1 |
Melting point: 387 K |
Boiling point: 457 K |
| isolated by Courtois in 1811 by treating the ash of seaweed with acid |
| astatine |
Atomic configuration: [Xe] 4f145d106s26p5 |
Atomic mass: (210) |
Pauling electronegativity: 2.2 |
Common oxidation states: ±,+3,+5,+7
|
Ionization energy: ~926 kJ mol-1 |
Electron affinity: ~270 kJ mol-1 |
Melting point: 575 K |
Boiling point: - |
| made by Corson, Mackenzie and Segr� in 1940 from 209Bi |
| The noble gases |
| elements with a full valence shell |
| Helium |
Atomic configuration: 1s2 |
Atomic mass: 4.0026 |
Pauling electronegativity: n.a. |
Common oxidation states: 0 |
Ionization energy: 2372 kJ mol-1 |
Electron affinity: 0
|
Melting point: cannot be frozen at atmospheric pressure
|
Boiling point: 4.2 K |
| identified in the solar chromosphere in 1868 by Lockyer and Frankland |
| Neon |
Atomic configuration: [He] 2s22p6 |
Atomic mass: 20.179 |
Pauling electronegativity: n.a. |
Common oxidation states: 0 |
Ionization energy: 2080 kJ mol-1 |
Electron affinity: 0
|
Melting point: 25 K
|
Boiling point: 27 K |
| isolated by distillation of air by Ramsay and Travers in 1898 |
| Argon |
Atomic configuration: [Ne] 3s23p6 |
Atomic mass: 39.948 |
Pauling electronegativity: n.a. |
Common oxidation states: 0 |
Ionization energy: 1520 kJ mol-1 |
Electron affinity: 0 |
Melting point: 84 K |
Boiling point: 87 K |
| isolated by Ramsay in 1895 from atmospheric nitrogen by adding Mg |
| Krypton |
Atomic configuration: [Ar] 4s24p6 |
Atomic mass: 83.80 |
Pauling electronegativity: 3.0 |
Common oxidation states: 0,+2 |
Ionization energy: 1351 kJ mol-1 |
Electron affinity: 0
|
Melting point: 116 K
|
Boiling point: 120 K |
| isolated by distillation of air by Ramsay and Travers in 1898 |
| Xenon |
Atomic configuration: [Kr] 5s25p6 |
Atomic mass: 131.29 |
Pauling electronegativity: 2.6 |
Common oxidation states: 0,+2,+4,+6 |
Ionization energy: 1170 kJ mol-1 |
Electron affinity: 0
|
Melting point: 161 K
|
Boiling point: 165 K |
| isolated by distillation of air by Ramsay and Travers in 1898 |
| Radon |
Atomic configuration: [Xe] 6s26p6 |
Atomic mass: (222) |
Pauling electronegativity: n.a. |
Common oxidation states: 0 |
Ionization energy: 1037 kJ mol-1 |
Electron affinity: 0
|
Melting point: 202 K
|
Boiling point: 211 K |
| observed as a short-lived radioactive element in 1902 by Rutherford and Soddy |
| Ununoctium |
Atomic configuration: [Rn] s27p6 |
Atomic mass: - |
Pauling electronegativity: - |
Common oxidation states: - |
Ionization energy: -
|
Electron affinity: -
|
Melting point: -
|
Boiling point: - |
| claimed in 1999 by team at Berkeley but withdrawn in 2001 |
| The lanthanoids |
| also known as the lanthanides and the Rare Earths |
| Cerium |
Atomic configuration: [Xe] 6s24f15d1 |
Atomic mass: 140.12 |
Pauling electronegativity: 1.12 |
Common oxidation states: +3, +4 |
Ionization energy: 541 kJ mol-1 |
Electron affinity: -
|
Melting point: 798 K
|
Boiling point: 3433 K |
| isolated in 1839 by the Swedish surgeon Mosander from the mineral ceria |
| Praseodymium |
Atomic configuration: [Xe] 6s24f3 |
Atomic mass: 140.91 |
Pauling electronegativity: 1.13 |
Common oxidation states: +3, +4 |
Ionization energy: 522 kJ mol-1 |
Electron affinity: -
|
Melting point: 931 K
|
Boiling point: 3520 K |
| isolated in 1885 by von Welsbach from the mineral ceria |
| Neodymium |
Atomic configuration: [Xe] 6s24f4 |
Atomic mass: 144.24 |
Pauling electronegativity: 1.14 |
Common oxidation states: +2, +3, +4 |
Ionization energy: 530 kJ mol-1 |
Electron affinity: -
|
Melting point: 1021 K
|
Boiling point: 3074 K |
| isolated in 1885 by von Welsbach from the mineral ceria |
| Promethium |
Atomic configuration: [Xe] 6s24f5 |
Atomic mass: - |
Pauling electronegativity: - |
Common oxidation states: +3 |
Ionization energy: 536 kJ mol-1 |
Electron affinity: -
|
Melting point: 1042 K
|
Boiling point: ~3000 K |
| first seen in 1947 by Marinsky, Glendenin and Coryell in U fission products |
| Samarium |
Atomic configuration: [Xe] 6s24f6 |
Atomic mass: 150.36 |
Pauling electronegativity: 1.17 |
Common oxidation states: +2, +3 |
Ionization energy: 542 kJ mol-1 |
Electron affinity: -
|
Melting point: 1345 K
|
Boiling point: 2076 K |
| isolated in 1879 by de Boisbaudram from the mineral ceria |
| Europium |
Atomic configuration: [Xe] 6s24f7 |
Atomic mass: 151.96 |
Pauling electronegativity: - |
Common oxidation states: +2, +3 |
Ionization energy: 547 kJ mol-1 |
Electron affinity: -
|
Melting point: 822 K
|
Boiling point: 1794 K |
| isolated in 1901 by Demarcay from the mineral ceria |
| Gadolinium |
Atomic configuration: [Xe] 6s24f75d1 |
Atomic mass: 157.25 |
Pauling electronegativity: 1.20 |
Common oxidation states: +3 |
Ionization energy: 595 kJ mol-1 |
Electron affinity: -
|
Melting point: 1313 K
|
Boiling point: 3273 K |
| isolated in 1880 by de Marignac from the mineral yttria |
| Terbium |
Atomic configuration: [Xe] 6s24f9 |
Atomic mass: 158.93 |
Pauling electronegativity: - |
Common oxidation states: +3, +4 |
Ionization energy: 569 kJ mol-1 |
Electron affinity: -
|
Melting point: 1365 K
|
Boiling point: 3230 K |
| isolated in 1843 by the Swedish surgeon Mosander from the mineral yttria |
| Dysprosium |
Atomic configuration: [Xe] 6s24f10 |
Atomic mass: 162.50 |
Pauling electronegativity: 1.22 |
Common oxidation states: +2, +3, +4 |
Ionization energy: 567 kJ mol-1 |
Electron affinity: -
|
Melting point: 1412 K
|
Boiling point: 2567 K |
| isolated in 1886 by de Boisbaudran from the mineral yttria |
| Holmium |
Atomic configuration: [Xe] 6s24f11 |
Atomic mass: 164.93 |
Pauling electronegativity: 1.23 |
Common oxidation states: +3 |
Ionization energy: 574 kJ mol-1 |
Electron affinity: -
|
Melting point: 1474 K
|
Boiling point: 2700 K |
| isolated in 1878 by Soret and Delafontaine from the mineral yttria |
| Erbium |
Atomic configuration: [Xe] 6s24f12 |
Atomic mass: 167.26 |
Pauling electronegativity: 1.24 |
Common oxidation states: +3 |
Ionization energy: 581 kJ mol-1 |
Electron affinity: -
|
Melting point: 1529 K
|
Boiling point: 2868 K |
| isolated in 1843 by Carl Mosander from the mineral yttria |
| Thullium |
Atomic configuration: [Xe] 6s24f13 |
Atomic mass: 168.93 |
Pauling electronegativity: 1.25 |
Common oxidation states: +2, +3 |
Ionization energy: 589 kJ mol-1 |
Electron affinity: -
|
Melting point: 1545 K
|
Boiling point: 1950 K |
| isolated in 1879 by Per Theodore Cleve from the mineral yttria |
| Ytterbium |
Atomic configuration: [Xe] 6s24f14 |
Atomic mass: 173.04 |
Pauling electronegativity: - |
Common oxidation states: +2, +3 |
Ionization energy: 603 kJ mol-1 |
Electron affinity: -
|
Melting point: 819 K
|
Boiling point: 1196 K |
| isolated in 1878 by de Marignac from the mineral yttria |
| Lutetium |
Atomic configuration: [Xe] 6s24f145d1 |
Atomic mass: 174.97 |
Pauling electronegativity: 1.27 |
Common oxidation states: +3 |
Ionization energy: 513 kJ mol-1 |
Electron affinity: -
|
Melting point: 1663 K
|
Boiling point: 3402 K |
| isolated (1907) by Urbain/James/von Welsbach from yttria |
| The actinoids |
| actinides: all elements after U are artifical |
| Thorium |
Atomic configuration: [Rn] 7s26d2 |
Atomic mass: 232.4 |
Pauling electronegativity: 1.3 |
Common oxidation states: +3, +4 |
Ionization energy: 587 kJ mol-1 |
Electron affinity: -
|
Melting point: 1750 K
|
Boiling point: 4788 K |
| isolated in 1828 by J. J. Berzelius from the mineral now known as thorite |
| Protactinium |
Atomic configuration: [Rn] 7s25f26d1 |
Atomic mass: 231.04 |
Pauling electronegativity: 1.5 |
Common oxidation states: +3, +4, +5 |
Ionization energy: 568 kJ mol-1 |
Electron affinity: -
|
Melting point: 1572 K
|
Boiling point: (4720 K) |
| identified in 1913 by Fajans and Gohring in 238U decay series |
| Uranium |
Atomic configuration: [Rn] 7s25f36d1 |
Atomic mass: 238.04 |
Pauling electronegativity: 1.38 |
Common oxidation states: +2, +3, +4, +5, +6 |
Ionization energy: 598 kJ mol-1 |
Electron affinity: -
|
Melting point: 1135 K
|
Boiling point: 3818 K |
| identified and isolated in 1789 by M. H. Klaproth in pitchblende |
| Neptunium |
Atomic configuration: [Rn] 7s25f46d1 |
Atomic mass: 237.05 |
Pauling electronegativity: 1.36 |
Common oxidation states: +3, +4, +5, +6, +7 |
Ionization energy: 585 kJ mol-1 |
Electron affinity: -
|
Melting point: 644 K
|
Boiling point: (3900 K) |
| made in 1940 by McMillan and Abelson by neutron bombardment of 238U |
| Plutonium |
Atomic configuration: [Rn] 7s25f6 |
Atomic mass: (244) |
Pauling electronegativity: 1.28 |
Common oxidation states: +3, +4, +5, +6, +7 |
Ionization energy: 605 kJ mol-1 |
Electron affinity: -
|
Melting point: 640 K
|
Boiling point: 328 K |
| made in 1940 by Seaborg, McMillan, Kennedy and Wahl from 238U |
| Americium |
Atomic configuration: [Rn] 7s25f7 |
Atomic mass: (243) |
Pauling electronegativity: 1.3 |
Common oxidation states: +3, +4, +5, +6 |
Ionization energy: 578 kJ mol-1 |
Electron affinity: -
|
Melting point: 1176 K
|
Boiling point: (2600 K) |
| made in 1944 by Seaborg, James, Morgan and Ghiorso from 239Pu |
| Curium |
Atomic configuration: [Rn] 7s25f76d1 |
Atomic mass: (247) |
Pauling electronegativity: 1.3 |
Common oxidation states: +3, +4 |
Ionization energy: 581 kJ mol-1 |
Electron affinity: -
|
Melting point: 1345 K
|
Boiling point: - |
| made in 1944 by Seaborg, James and Ghiorso from 239Pu |
| Berkelium |
Atomic configuration: [Rn] 7s25f86d1 |
Atomic mass: (247) |
Pauling electronegativity: 1.3 |
Common oxidation states: +3, +4 |
Ionization energy: 601 kJ mol-1 |
Electron affinity: -
|
Melting point: 1050 K
|
Boiling point: - |
| made in 1949 by Seaborg, Ghiorso and Thompson from 241Am |
| Californium |
Atomic configuration: [Rn] 7s25f10 |
Atomic mass: (251) |
Pauling electronegativity: 1.3 |
Common oxidation states: +3, +4 |
Ionization energy: 608 kJ mol-1 |
Electron affinity: -
|
Melting point: 900 K
|
Boiling point: - |
| made in 1944 by Street/ Seaborg/ James / Ghiorso from 242Cm |
| Einsteinium |
Atomic configuration: [Rn] 7s25f11 |
Atomic mass: (252) |
Pauling electronegativity: 1.3 |
Common oxidation states: +3 |
Ionization energy: 619 kJ mol-1 |
Electron affinity: -
|
Melting point: 860 K
|
Boiling point: - |
| identifed in 1952 at Berkeley in thermonuclear explosion debris |
| Fermium |
Atomic configuration: [Rn] 7s25f12 |
Atomic mass: (257) |
Pauling electronegativity: 1.3 |
Common oxidation states: +3 |
Ionization energy: 627 kJ mol-1 |
Electron affinity: -
|
Melting point: 1527 K
|
Boiling point: - |
| identifed in 1952 at Berkeley in thermonuclear explosion debris |
| Mendelevium |
Atomic configuration: [Rn] 7s25f13 |
Atomic mass: (258) |
Pauling electronegativity: 1.3 |
Common oxidation states: +3 |
Ionization energy: 635 kJ mol-1 |
Electron affinity: -
|
Melting point: 827 K
|
Boiling point: - |
| made in 1955 by Seaborg, Ghiorso, Harvey, Choppin and Thompson from 253Es |
| Nobelium |
Atomic configuration: [Rn] 7s25f14 |
Atomic mass: (259) |
Pauling electronegativity: 1.3 |
Common oxidation states: +2, +3 |
Ionization energy: 642 kJ mol-1 |
Electron affinity: -
|
Melting point: 827 K
|
Boiling point: - |
| made in 1956 at Dubna from 15N bombardment of 243Am |
| Lawrencium |
Atomic configuration: [Rn] 7s25f146d1 |
Atomic mass: (260) |
Pauling electronegativity: 1.3 |
Common oxidation states: +3 |
Ionization energy: -
|
Electron affinity: -
|
Melting point: 1627 K
|
Boiling point: - |
| made in 1961-71 at Berkeley and Dubna from Cf and 243Am |
 |
||||||
|
||||||