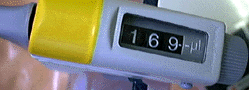

Stoichiometry
This module reviews the units and techniques chemists use to quantitatively describe chemical processes.
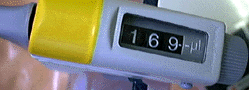 |
 |
|||||||||||||||
Stoichiometry |
||||||||||||||||
| Accurate
measurement was a turning point in the development of chemistry as we know
it. Careful measurements of mass and volume were the foundation for the
atomic theories of matter, leading to our modern molecular pictures for
all chemical phenomena - in the laboratory, in industry, in biology, or
in the environment. This module reviews the units and techniques chemists use to quantitatively describe chemical processes. |
||||||||||||||||
|
|
||||||||||||||||
|
|
||||||||||||||||